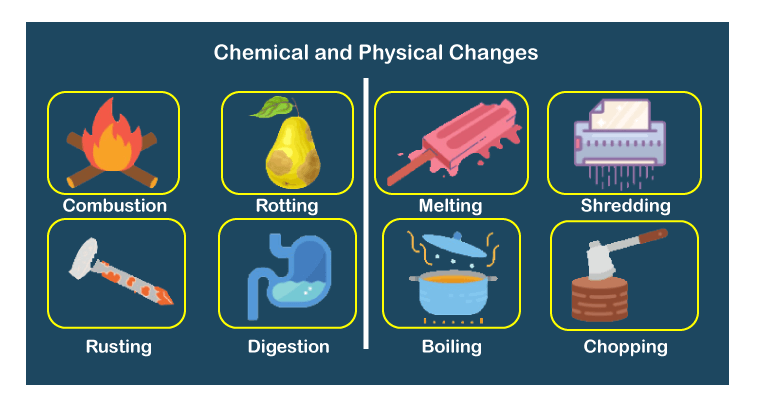
Because we are changing the bonding, this means we are also changing how much of this "stored energy" there is in a reaction. After the reaction, the matter's chemical composition changes, and a new substance forms. During this reaction, bonds between atoms break or new ones form. Chemical bonds have potential energy or "stored energy". A chemical change occurs when a reaction causes a change at a molecular level. Physical change is reversible, meaning the original state can be restored, while chemical change is usually irreversible. Most chemical changes, on the other hand, take longer time to become discernible. Physical change occurs faster and sometimes instantaneously. Another big difference between physical and chemical change is the speed of transformation. The bonds in the reactants are broken and the bonds of the products are formed. Physical change absorbs energy in the form of heat or light, whereas chemical change requires an input of energy (activation energy). You can not ‘un-burn’ ash to transform it back to paper. Remember that all chemical reactions involve a change in the bonds of the reactants. When liquid water becomes ice, energy is released. 
The differences between the two types of change. For example, when solid dry ice vaporizes (physical change), carbon dioxide molecules absorb energy. These physical changes are often changes in state such as melting, freezing, boiling, condensing and dissolving. Endothermic reaction: surroundings get cooler and delta H is greater than 0, Exothermic reaction: surroundings get warmer and delta H is less than 0ĭuring phase changes, energy changes are usually involved. The difference between a physical reaction and a chemical reaction is composition.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
